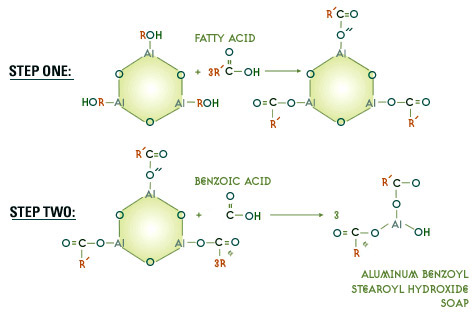
The general reaction
Typical Formulation
More efficient soaps are made with acids ratios of benzoic to fatty acids of less than one. Assuming the following starting parameters:
A 650 SUS solvent neutral base stock, with an aniline point of 99°C, a 7.5% soap, with benzoic to fatty acid mol ratio of 0.75, and a total acids to aluminum ratio of 1.9, a typical formulation might be as follows:
The facts and recomendations made herein are based on our research and the research of others, and while they are believed to be accurate, they are intended for use by skilled persons at their own risk. We assume no liability for the events resulting or damages incurred from the use of this information. Statements concerning the possible use of our products are not intended as recommendations to use them in the infringement of any patent.